March 2026
Catalysts
The octane boost: How FCC chemistry and ingenuity fill the last barrel of the alkylation unit
In today’s refining landscape, the pursuit of premium gasoline octane is more than a performance metric—it is a profitability driver. Refiners must use innovative strategies to meet the growing economic octane incentive driven by stringent fuel economy standards and high-performance engines. To stay ahead, leading refiners are embracing synergies of new chemistry and operating strategies. This article explores how Marathon Petroleum and BASF have partnered to optimize fluid catalytic cracking (FCC) and alkylation integration, leveraging advanced FCC catalyst technology and operational ingenuity to maximize high-octane gasoline, resulting in > $1/bbl benefit.
The octane incentive: A rising tide. The octane incentive has steadily climbed over the past decade, driven by:
- Tightening environmental regulations to reduce sulfur in gasoline, which requires more severe hydrotreating, resulting in octane loss. This includes the U.S. Environmental Protection Agency’s (EPA’s) Tier3 sulfur gasoline reduction that reduced sulfur content in gasoline from 30 parts per million (ppm) to 10 ppm starting in 2017, with a compliance period from 2017–2019.
- Increasing fuel economy standards. This includes the U.S. Corporate Average Fuel Economy (CAFE) regulations, pushing automakers toward higher compression engines requiring premium fuel.
- Increased the processing of shale oil feeds (also known as light tight oils). Shale oils naturally yield lower octane gasoline compared to traditional crude oils.
According to U.S. Energy Information Administration (EIA) data, the spread between premium and regular unleaded gasoline continues to widen. As of January 2026, the premium-to-regular gasoline spread was $0.91/gal (FIG. 1). This economic incentive has elevated high-octane alkylate, typically ranging from 90 octane–95 octane, to one of the most valuable refinery streams. Alkylate is produced by reacting light olefins with isobutane in the presence of an acid catalyst, such as sulfuric or hydrofluoric acid. The light olefin mix is primarily propylene and butylenes, although alkenes (C5=) can be used, as well.

FIG. 1. U.S. EIA weekly retail gasoline prices premium. Regular differential is in $/gal. Source: U.S. EIA.
FCC + alkylation: A synergistic value chain. The FCC unit (FCCU) takes heavy oil and produces a range of products from distillate to light olefins. The FCCU contributes roughly half of a refinery’s gasoline pool and serves as the primary source of light olefins for alkylation. FCCUs produce both propylene (C3=) and butylenes (C4=), with the flexibility to selectively favor one over the other via catalyst chemistry and operational adjustments. This article will review the most profitable feed composition for the alkylation unit (C3= or C4=) to determine how to optimize the FCCU.
Eqs. 1 and 2 are simplified sulfuric alkylation chemistry. These numbers will vary with the mixed butylenes (1-butene, cis-2-butene, trans-2-butene and isobutene), the isobutane-to-olefin ratio (I/O ratio), contaminants and temperature. In other words, for a specific unit, mileage may vary. While mileage may vary, this gives a good understanding of what is more valuable to feed the alkylation unit: C3= or C4=.
Simplified sulfuric acid alkylation chemistry:
1 bbl C4= + 1.15 bbl iC4 → 1.78 bbl alkylate @ 95 research octane number (RON) (1)
1 bbl C3= + 1.30 bbl iC4 → 1.68 bbl alkylate @ 90 RON (2)
These reactions demonstrate that butylene alkylate offers superior octane, a higher yield and lower consumption of iC4 over propylene. C4= alkylation requires less acid consumption, reducing operating costs. Another operational advantage of C4=s is the lower exotherm generated from the alkylation reaction compared to C3=s. C3= alkylation has about 35% higher heat of reaction than C4=, requiring more refrigeration duty. Hydrofluoric acid alkylation units also value C4=s over C3=, but the differential is less due to technological differences.
FIG. 2 illustrates the estimated value incentive of alkylating C4= over C3= as a function of octane.

FIG. 2. Sulfuric acid alkylation C4= value over C3= value vs octane. The calculation assumes gasoline at $70/bbl and iC4 at $20/bbl.
For example, if a refinery has zero octane incentive, then C4= should be 25% higher than C3=, not accounting for acid cost or refrigeration limits. If a refinery has $1.50/bbl octane incentive, then C4= are valued 60% above C3=. This shows C4= are the preferred feedstock an FCCU should make for alkylation.
When evaluating FCC economics, consideration must be given for alkylation limits tied to the FCCU. Most FCCUs have undersized gas plants due to rate expansion over the years, which may limit separation of light products. Aan undersized gas plant will limit light olefin recovery. Alkylation units have propane rejection limits, where less propane produced by the FCCU means more capacity utilization of the alkylation unit.
FCC optimization: From conventional to modern strategies to maximize C4= production for alkylation. Historically refiners have used two traditional FCC strategies to fill the alkylation unit:
- ZSM-5 additive: Increases total light olefins and octane, but produces incrementally more C3= than C4=, along with some ethylene and iC4.
- Raising riser outlet temperature (ROT): Increases light olefins and octane but also increases dry gas and saturates (straining gas plant capacity) and uses more air.
Modern FCC approaches include:
- Using advanced catalyst technology to increase light olefins production, with increased selectivity towards C4= over C3= production
- Using catalysts and operating moves to lower light saturate production (light saturates are one of the lowest valued FCC products)
- Improved catalyst delta coke and reduced operating severity—fewer pounds of coke means more liquid yield
- Improved slurry destruction to allow lower ROT operation (lowers saturate production).
For advanced catalysts, the co-author’s company’s multiple framework topology (MFT) catalysts combine multiple zeolite frameworks with an advanced matrix. Currently, there are four catalysts based off this platform designed for different feed types and yield objectives. This family of catalysts delivers:
- Higher C4= yield
- Improved butylene-to-propylene selectivity
- Improved liquified petroleum gas (LPG) olefinicity
- Enhanced FCCU naphtha octane
- Lower coke yield and saturates.
CASE STUDY: MARATHON UNIT A
Marathon Unit A is a landlocked refinery processing hydrotreated vacuum gasoil (VGO) feed and has no gasoline sulfur post-treater. The unit has historically used olefin additives to maximize alkylation feed. The refinery increased the hydrotreater severity over the years to comply with Tier 3 regulations, resulting in octane loss (FIG. 3).

FIG. 3. Unit A’s FCCU feed sulfur decreasing over the years due to increased hydrotreating to meet Tier 3 gasoline sulfur specifications.
Unit A has a strong economic incentive to increase the octane of the gasoline pool. To address the economic incentive, the unit transitioned to the co-author’s company’s first-generation FCC catalysta, and later to the latest-generation MFT FCC catalystb for increased alkylate production and higher FCC gasoline octane. The catalyst changes increased C4= yield sustainability over the years.
In addition to the catalyst change, the unit was able to operate at a lower ROT (FIG. 4), which allows for less pounds of coke produced, less air and less low-value LPG saturates like propane. FIG. 4 also shows the reductions in saturates, which are beneficial for economics, alleviate the constrained gas plant and send less propane to the alkylation unit. Note: In recent years, the saturate yield increased due to condensate processing.

FIG. 4.Unit A’s ROT decreased over the years through a change in catalyst. The catalyst changes and lower ROT resulted in lower light saturates (propane, n-butane, i-butane). The catalyst periods are shown along with the transition between catalysts.
FIG. 5 shows the propylene and butylene yield over the catalyst change. Adjusting the catalyst and operating changes, propylene was constant while butylenes increased by 2.5 vol%. These shifts would not have been attainable with the conventional strategies discussed above, as those would have increased propylene and isobutane.

FIG. 5. Unit A’s propylene and butylene production. Through the catalyst changes, propylene production remained constant, while C4= increased by 2.5 vol%.
As a result of the change to the first-generation FCC catalysta, the unit achieved a profit improvement of $0.50/bbl through lower saturates, higher LPG olefins, higher octane and improved delta coke.
When the latest-generation MFT FCC catalystb was utilized in the unit, profitability increased to $0.71/bbl through increased butylene yield, a higher C4=/C3= ratio and lower delta coke.
CAST STUDY: MARATHON UNIT B
Like Unit A, Marathon Unit B also processes hydrotreated VGO and has no gasoline sulfur post-treater. Under Tier 3 regulations, Unit B experienced a 2 RON–3 RON loss due to increased hydrotreater severity (FIG. 6). By using the co-author’s company’s first-generation and latest-generation FCC catalystsa,b, the unit was able to fill the alkylation unit more profitability and remove the olefins additive the unit was utilizing.
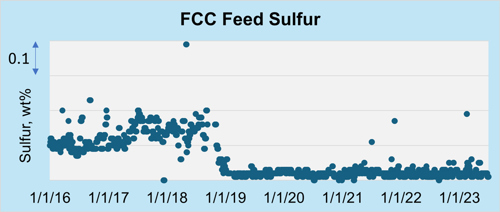
FIG. 6. Unit B’s FCCU feed sulfur decreasing over the years due to increased hydrotreating to meet Tier 3 gasoline sulfur specifications.
FIG. 7 highlights the large increase in butylene selectivity and the increase in the gasoline’s RON achieved through the catalyst changes.

FIG. 7. Unit B increase in butylene selectivity and FCCU gasoline RON.
The first-generation FCC catalysta improved unit profitability by $0.40/bbl through lower LPG saturates, higher LPG olefins and higher gasoline octane. The latest-generation FCC catalystb further increased profitability by $1.40/bbl through increased LPG olefins, with significantly higher butylene selectivity and achieving filling the alky and allowing the unit to back out olefins additives like ZSM-5. The catalyst change impact on the alkylation unit included more alkylate feed, especially more C4= to produce high-octane alkylate. In 2022, an alkylation unit revamp increased the rate with contactor bundle upgrades. The contactor temperature was maintained with the rate increase, and the FCCU met the demand for higher light olefins.
Takeaway: Filling the last barrel with value. The octane incentive is not only a long-term trend, but also one of the defining success metrics of modern refining economics. Refiners can capitalize on this trend by selectively filling the alkylation unit with butylenes to produce premium gasoline as opposed to other less effective light olefins, saving on operating costs and improving alkylate yields. The integration of FCC and alkylation is no longer optional—it is essential. Optimizing the FCCU or alkylation unit on its own is not a game-winning strategy: both must work together to drive optimum refinery operation. This is applicable for all refineries that have an FCCU and alkylation unit, regardless of the type of alkylation unit and FCCU design. By combining advanced catalyst technology with strategic operational moves, refiners can unlock significant value in the gasoline pool. The collaboration between the co-authors’ companies exemplifies how chemistry and ingenuity can fill the last barrel—not just with product, but with proven profitability of > $1.00/bbl, which is an indication of a more resilient, more competitive and more profitable refining operation.
NOTES
a BASF Fourte®
b BASF Fourtiva™








Comments